I wanted to share more about our failed food challenge from last month. But in order to do that, we need to backtrack, because I never fully shared what we were doing with the Peanut Patch to get to this point.
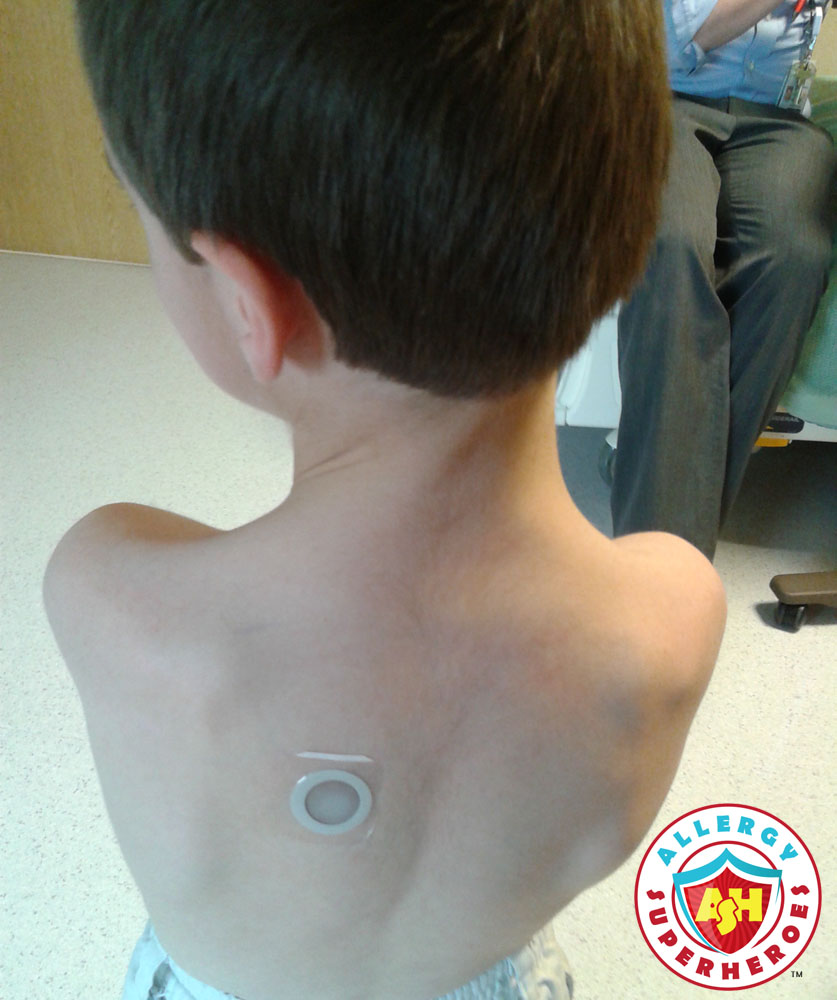
This photo was taken on February 28, 2017, when my little guy Kal was still in kindergarten. This was the first patch he ever wore for the Viaskin REALISE Peanut Patch Study.
Like a lot of people, I was extremely excited about the new Peanut Patches being developed by DBV technologies. When I learned that our local research hospital was going to participate in the latest patch trial, I harassed checked in with them regularly to make sure they knew we wanted to participate. My older son Zax had already completed one study at National Jewish and was finishing up a second. That inside relationship probably helped me secure Kal a spot in REALISE.
Patch technology was exciting because it promised to help an allergic child achieve desensitization by wearing a daily patch on the skin, rather than by ingesting the allergen. This branch of desensitization is called Epicutaneous Immunotherapy, or EPIT.
What is Immunotherapy?
In layman’s terms, EPIT is a therapy that introduces allergens to the immune system through the skin. You can think of them in terms of nicotine patches, except that the idea is to wean the patient ON to peanut (or other allergens) rather than weaning them OFF.
As the body begins to tolerate the allergen, the patient is considered desensitized. This means they can tolerate larger amounts of allergens before their body triggers a reaction, or that any reaction they have will be less severe. Or both.
Other methods of desensitization existed, but were hard to come by, and all involved ingesting the allergen to some extent. Oral Immunotherapy (OIT) involves eating progressively larger doses of allergen on a daily basis. Sublingual Immunotherapy (SLIT) involves placing drops of allergen under the tongue. Subcutaneous Immunotheraphy (allergy shots) are injected immunotherapy. Allergy shots have been around for a long while, but aren’t safe to do for food allergies.
How the Peanut Patch works
But back to the Patch. There’s a small amount of peanut protein (250 micrograms) on each patch. The patch adheres to the skin, creating a moisture-proof barrier. The allergens dissolve in the patient’s own sweat, which gets trapped under the patch. The sweat is then reabsorbed through the skin and taken directly to the lymphatic system, bypassing the digestive tract and the bloodstream entirely. In this way, the body begins to recognize and tolerate the allergen without having to ingest it.

To my understanding, OIT is a big commitment. It involves activity restrictions before and after dosing, and is often fraught with side effects and reactions.
By contrast, I loved the sound of EPIT. There were no activity restrictions, and all you had to remember was “patch on, patch off.” While OIT promises to make a patient’s allergic threshold higher, the patch promises to be easy and minimally invasive.
REALISE
I was thrilled to enroll Kal in REALISE. REALISE was a Phase III clinical trial for Viaskin Peanut. It was considered a “safety study.” While the FDA had granted DBV “fast-track” status for their therapy, the FDA still wanted more data on how the peanut patch would be used in clinical practice. As such, Kal didn’t have to do any food challenges for the study. As long as he had had a reaction in the past and not just a diagnosis via testing (he had), then we were eligible to screen.
For the first 6 months of the study, it was blind. That means that we didn’t know whether Kal had real peanut patches on his back, or placebo. After six months the trial became “open label,” which means everyone was on real peanut patches and knew it. We later learned that Kal started with placebo.
Everyone enrolled in Viaskin REALISE would receive three years of active patch treatment. That meant that we were in the trial for 3 1/2 years (6 months on placebo, and then 3 years on active patches.)
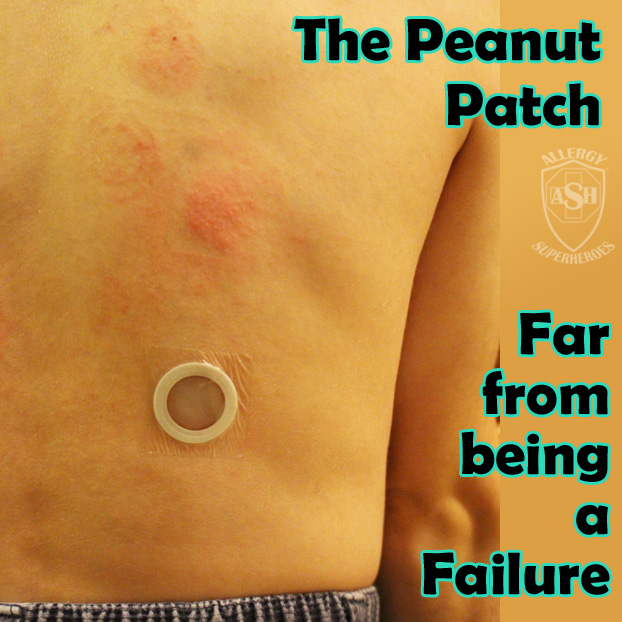
It started out great. Once on active treatment, Kal did experience some discomfort at the patch site (itching, redness, and swelling.) These side effects diminished over time. And honestly, they’re a small price to pay for potential allergen tolerance!

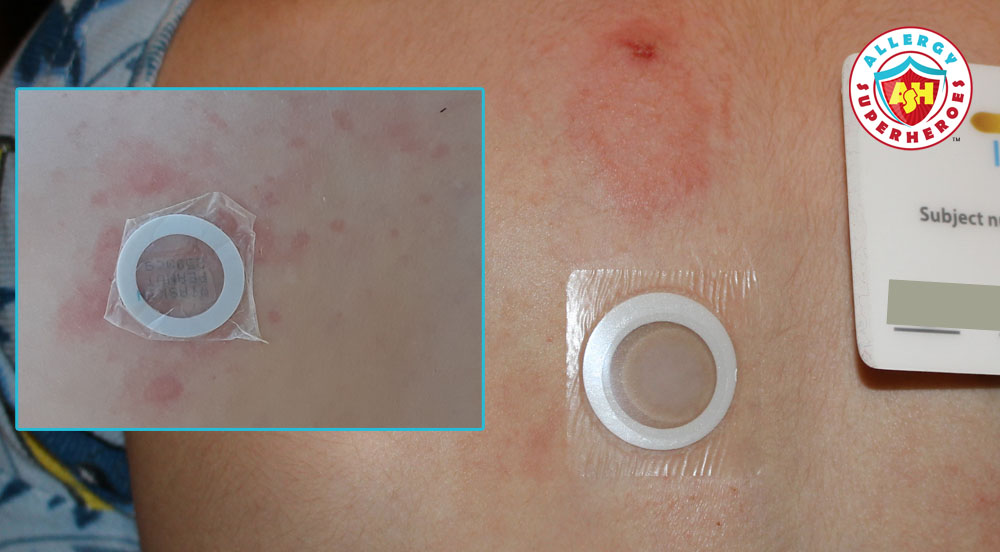
Bumps in the Road
For the first 18 months, the patches stuck to Kal’s skin very well. When it came time to take them off, we had to scrape up a corner with a fingernail until we had enough to grip. Removing them was difficult for a little kid at first, but eventually he understood that ripping it off quickly (like a Band-Aid) was the easiest and fastest way.
We got new patches every 6 months, with each medical visit. And at the 18 months mark (12 months active), things began to go downhill.
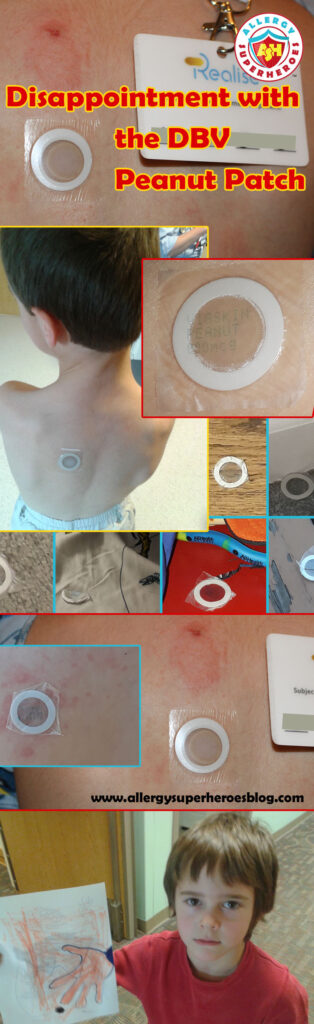
At that time, the patches stopped sticking. They would go on okay, but the adhesive seemed to be weaker. They would fall off early in the day, or would migrate to a lower spot on his back.
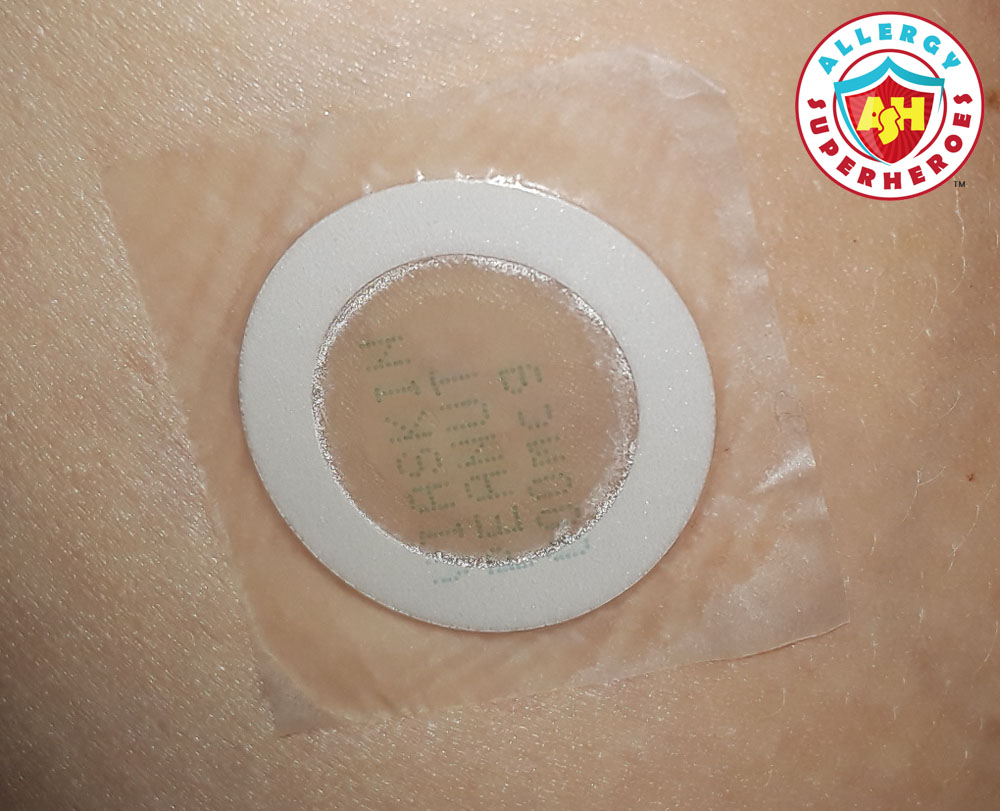
I was worried that I’d messed them up somehow. At the time, we were supposed to store them in the refrigerator. I was worried that they’d gotten too hot on the way home, and that I’d destroyed 6 months of expensive experimental patches.
I soon learned that we were not the only family experiencing problems, though. Other people were also complaining that the patches had stopped sticking. We dutifully continued to apply them, and tried to approximate when they fell off in our Study Log. But we began to be concerned–how long would a patch need to stay on in order to be effective?
(DBV removed the refrigeration requirement during our final year. Now they simply need to be kept at or below room temperature.)

No response to concerns
Our study nurse regularly submitted complaints to DBV. Every 6 months we got new patches, and sometimes we were told that DBV had adjusted the adhesive. However, we never noticed a change for the better. If anything, patch adhesion continued to get worse.
By the end of the trial, I only removed a patch every month or two. The rest fell off on their own. We never really knew when they fell off, but estimated it stayed on for 12 hours or fewer most days. I was always finding patches on the ground, on clothes, on blankets, and in the most random of places.
(We ended the study on August 11, 2020. I continued finding lost patches through August, and even found a few in September and October!)

Peanut Patch not FDA Approved
Considering our experience, it was no surprise when we learned, in August of this year, that the FDA did not approve Viaskin Peanut. The FDA did not have any safety concerns, but they “identified concerns regarding the impact of patch-site adhesion on efficacy and indicated the need for patch modifications, and subsequently a new human factor study.”
In other words, the patch didn’t stick for lots of participants. The FDA had concerns on how effective it was when in place for only a few hours. They wanted modifications to make it work properly, and a new study to prove that the modifications work.
Early on in the study, I joined an online community of patch participants. Through them, I learned that many other people had experienced adhesion problems, though not everyone experienced them at the same time. What worked for one patient didn’t necessarily work for another. This made me wonder if skin types made a difference?
Adhesive for the Peanut Patch
This is why I think that to be successful, DBV should release their patches with multiple adhesive options. I thought this even before the FDA ruling. I feel that with this solution, doctors could start by prescribing the adhesive that works for most people, and then switch to other adhesives if the first one doesn’t stick.
But what do I know? I’m just a parent with a child participant in one of their peanut patch studies. Unfortunately, I don’t have any sway (or voice) with DBV.
I made my multiple-adhesive suggestion to the study nurse and doctor at our research site, and asked them if they could pass it on. They kinda laughed bitterly and then told me that DBV doesn’t listen to them, either.
This is an unfortunate trend that I’ve also seen verified by my online patch community. DBV tends to brush off complaints and suggestions made by study employees and participants. The chief example is the “goop.”

My son was spared dealing with the “goop,” as REALISE didn’t involve any food challenges. But for people in other trials, the refrain was the same. The “goop” was horrible.
What is the “goop?”
The “goop” is DBV’s way of conducting food challenges. All challenges have to be “Double Blind Placebo Controlled” (DBPC.) That means that the patient does two challenges: one to peanut and one to a placebo. And nobody present knows which day is peanut and which is placebo until after both challenges are over. This way, they can make sure patients are really reacting and not just thinking they feel symptoms because of anxiety.
DBPC challenges are standard in food allergy trials, but DBV didn’t rely on research sites to mix their own trial foods. They standardized the food participants ate in the form of the “goop.”
From the way others have described it, the “goop” was this sticky, gelatinous substance with the consistency of a booger. Participants generally found it disgusting and difficult to choke down. More than one member of the online community said their child dropped out of the study because of the goop. Many have speculated that the “goop” negatively affected DBV’s food challenge numbers when it made kids throw up or refuse to go on.
Our research nurse said that when they were overseeing a trial that involved goop, she sent numerous complaints to DBV. Her patients universally hated the goop and wanted something else. Complaints poured in from other study sites too.
DBV responds
DBV eventually responded to their study population as a whole. They said that the “goop” is similar to a popular French candy (DBV is based in France) and that European kids love it. Our nurse said they basically finished by saying that American kids have unrefined palates.
Not only is this response rather insulting and dismissive, it’s actually quite unprofessional. I don’t know how many participants came from which countries, but I do know that the U.S. has a very large population compared to Europe. If I were to guess, I’d imagine that more than 50% of participants probably come from the U.S.
Ignoring the fact that a large number of participants can’t stand their challenge food is dismissive. But they are also ignoring the fact that the “goop” could impact their results.
Many participants may have prematurely ended their food challenges because of the “goop.” Gagging on it or throwing it up could be mistaken for symptoms. Refusal to finish the doses despite a lack of symptoms sometimes ended in prematurely exiting a study–but also meant the participant’s tolerance couldn’t be measured. All of this meant DBV probably didn’t have an accurate picture of how well their product works.
Even though we didn’t have to deal with the “goop,” we saw other examples of DBV’s dismissiveness in REALISE. Our nurse told us about the number of complaints she submitted on our behalf, once the patches stopped staying on. She submitted complaints for some of her other patients, too. All she ever got in return was a generic “received” reply. And only once did our new patches come with an assurance that they had a “better” adhesive.
Gradual disappointment
Participation did get rather frustrating, over time. We went from general enthusiasm to concern that the patches weren’t in place long enough to do their job. We felt that our adhesive issues were being ignored. The shiny and promising new technology lost its luster and turned into something I’m not sure I would be willing to spend money on.
After all, estimates have indicated that the peanut patch could be expensive. And the question of whether insurance will cover it is still unanswered. Why would I shell out thousands of dollars for a product that doesn’t stick? If it doesn’t stick–will it work?
It was doubly frustrating because the patches had stayed on before. It could work. I wanted to like this product, and was disappointed that I no longer did.
Future of the Peanut Patch
I was enthused when the Peanut Patch was making headlines for being fast-tracked by the FDA. Now, I think the FDA was right to send DBV back for more work. I think they need to replace the “goop” in any study that involves food challenges.
I think they also need to make adhesive a variable in their next trial. They should consider having more than one adhesive available for allergists and pharmacies to order.
And most importantly, they should learn to listen. Listen to the participants trying their product, and to the doctors and nurses coordinating them. Perhaps DBV’s product would be farther along in the approval process if they’d been listening already.
Please check out my next post to see what happened to Kal when REALISE ended.




So sorry that Kal had a negative experience with what could have been a valuable trial had DBV listened to feedback. That said, thanks for such a terrific description.
Thanks. We really had high hopes going in. It’s so frustrating, because I still think it would be a great option if it stayed on properly. I really love the unobtrusive nature of gaining tolerance. I want to like it, but by the time they finally get their act together, my kids are going to be too old to have it prescribed. (I’m pretty sure they’re looking at a max of 12, initially.)